TTV Risk Assessment Tool
TTV Risk Assessment Tool
This tool estimates your immune risk based on Torque Teno Virus (TTV) levels. TTV monitoring provides a more complete picture of your immune function than drug levels alone.
When someone takes immunosuppressive drugs-whether after a kidney, liver, or heart transplant, or for autoimmune conditions like lupus or rheumatoid arthritis-they’re walking a tightrope. These drugs prevent the body from attacking its own organs or a transplanted organ, but they also lower the body’s ability to fight off infections and can damage the kidneys, liver, or bones. That’s why monitoring during immunosuppressive therapy isn’t optional. It’s the difference between staying healthy and ending up back in the hospital.
Why Monitoring Isn’t Just a Routine Checkup
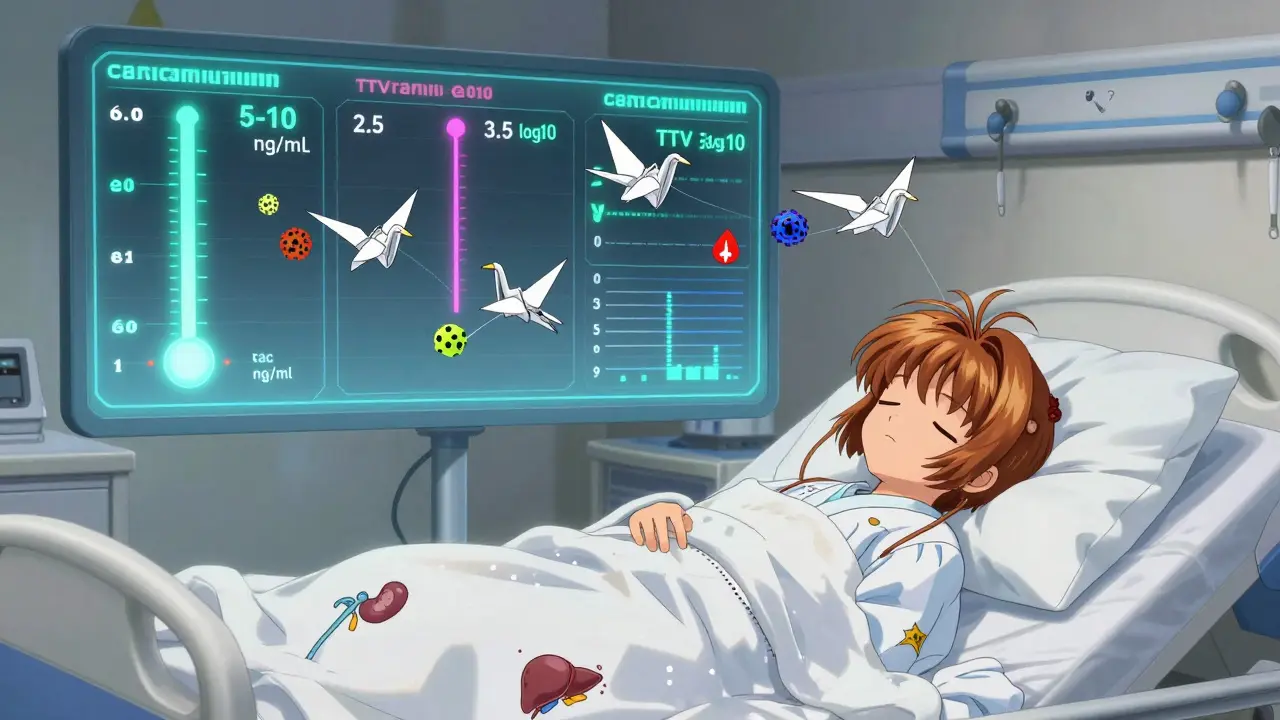
Not all immunosuppressants are the same. Some, like corticosteroids (prednisone), have predictable effects and don’t need constant blood checks. Others, like tacrolimus or cyclosporine, have a razor-thin line between helping and harming. The difference between the right dose and a toxic one can be as small as 2-4 times. Two people on the same pill dose can have completely different blood levels-one might be perfectly protected, the other at risk of rejection or kidney damage. That’s why therapeutic drug monitoring (TDM) is the backbone of care. For drugs like tacrolimus, the target range is usually 5-10 ng/mL in the first three months after transplant, then lowered to 3-7 ng/mL. Too low? Rejection risk jumps. Too high? Kidney damage, tremors, or even seizures can happen. Cyclosporine has a similar story, with targets around 100-200 ng/mL. But here’s the catch: these levels change daily based on diet, other medications, and even your body’s metabolism. That’s why checking them regularly isn’t bureaucracy-it’s survival.What Lab Tests Are Done and Why
Beyond drug levels, routine blood tests paint the full picture. Every 1-3 months, patients get a panel that includes:- Complete blood count (CBC): To catch low white blood cells (leukopenia), anemia, or low platelets-all common with mycophenolate or sirolimus.
- Creatinine and electrolytes: To track kidney function. Cyclosporine and tacrolimus can raise creatinine by 30% or more in a quarter of patients.
- Liver enzymes: To spot early signs of liver stress from drugs like sirolimus.
- Calcium, magnesium, phosphate: Cyclosporine often causes magnesium levels to crash, leading to muscle cramps or heart rhythm problems.
- Fasting glucose and lipids: Tacrolimus increases diabetes risk by 30%, and sirolimus can spike cholesterol in 60-75% of patients.
Imaging: More Than Just a Picture
Blood tests show what’s happening inside the bloodstream. Imaging shows what’s happening in the organs. A routine yearly renal ultrasound checks for scarring, blockages, or reduced blood flow to the transplanted kidney. If a patient has a cough or shortness of breath, a chest X-ray can catch pneumonitis-a rare but dangerous side effect of sirolimus that looks like pneumonia on imaging. For those on long-term steroids, bone density scans (DXA) are critical. After just one year of steroid use, bone mineral density can drop by 10-15%. That’s why annual scans start after year one. Catching osteoporosis early means adding calcium, vitamin D, or even bisphosphonates before a hip or spine fracture happens.The New Frontier: TTV as an Immune Meter
A breakthrough is emerging-not from a new drug, but from a virus. Torque Teno Virus (TTV) is harmless in healthy people. But in transplant patients, it multiplies like crazy when the immune system is suppressed. Scientists realized: the more TTV in the blood, the weaker the immune system. Studies show a clear sweet spot: a TTV level between 2.5 and 3.5 log10 copies/mL. Below that? Rejection risk triples. Above that? Infection risk doubles. In a major trial called TTVguideIT, patients whose drug doses were adjusted based on TTV levels had 28% fewer infections and 22% fewer rejections than those on standard monitoring. This isn’t science fiction-it’s happening now in leading transplant centers. The beauty? TTV testing is simple. Just a blood draw. No special equipment. It’s cheaper than LC-MS/MS drug testing and gives a real-time read on immune function, not just drug concentration. It’s like having a dashboard that tells you not just how much gas is in the tank, but how hard the engine is working.
The Reality: Cost, Access, and Human Burden
Not every hospital can afford this. Liquid chromatography-tandem mass spectrometry (LC-MS/MS), the gold standard for measuring tacrolimus and sirolimus, costs $150-$250 per test. Immunoassays are cheaper ($50-$100) but can give false readings due to cross-reactions with metabolites. Many clinics still use the cheaper method because of budget limits. Patients face real burdens too. In the first year after transplant, it’s common to have 12-18 blood draws. Some patients avoid appointments because they’re anxious or overwhelmed. One study found 35% of transplant recipients reported significant stress around lab visits. And in 68% of U.S. transplant centers, protocols vary between kidney, liver, and heart teams-no standardization means inconsistent care. The best-run centers have dedicated immunosuppression teams: pharmacists, nurses, and physicians who review every result within 24 hours. They adjust doses before problems arise. They explain the numbers to patients. They make monitoring feel like support, not surveillance.What’s Next: AI, Point-of-Care Tests, and Non-Invasive Monitoring
The future is already here in pilot form. A 2023 AI model analyzed years of lab data, TTV levels, and drug concentrations-and predicted acute rejection 14 days before symptoms appeared with 87% accuracy. That’s game-changing. Point-of-care devices are in development. Imagine a handheld machine at your clinic that gives tacrolimus levels in 15 minutes, like a glucose meter. FDA approval is expected by 2026-2027. Even more futuristic? Exhaled breath tests for immunosuppressant metabolites. Early animal studies show promise. No needles. Just a breath into a device. The bottom line? Monitoring during immunosuppressive therapy has evolved from guesswork to precision medicine. It’s not about doing more tests-it’s about doing smarter ones. TTV, AI, and better lab methods are turning reactive care into proactive protection. For transplant patients and those with autoimmune diseases, this isn’t just science. It’s peace of mind.Why do some immunosuppressants need blood tests and others don’t?
Drugs like tacrolimus, cyclosporine, sirolimus, and mycophenolate have a very narrow range between being effective and being toxic. Small changes in dose can cause rejection or organ damage, so blood levels must be tracked. Corticosteroids like prednisone and newer drugs like belatacept have more predictable effects and don’t require routine monitoring.
How often do I need blood tests after a transplant?
In the first three months, you’ll likely have blood drawn every 1-2 weeks. After that, it usually drops to every 2-4 weeks for the next 6-12 months. By year one, most patients are tested monthly. After the first year, testing typically slows to every 2-3 months unless there’s a problem. TTV monitoring may eventually reduce this frequency by giving longer-term immune insight.
Can I stop taking my immunosuppressants if my levels look good?
Never. Even if your drug levels are perfect and your labs look normal, stopping immunosuppressants can trigger immediate rejection of your transplant or a flare-up of autoimmune disease. Monitoring helps adjust doses, not eliminate them. Always follow your transplant team’s instructions.
What’s the point of TTV testing if I’m already getting drug levels checked?
Drug levels tell you how much medicine is in your blood. TTV tells you how your immune system is actually responding. Two people can have the same tacrolimus level but very different TTV levels-one might be at high risk of rejection, the other at high risk of infection. TTV gives a functional readout, not just a chemical one.
Is TTV testing available everywhere?
Not yet. TTV monitoring is still being validated in large trials and isn’t standard in most clinics. It’s currently offered in major transplant centers in the U.S., Canada, Europe, and Australia as part of research programs. Widespread use is expected after FDA clearance in 2025-2026.








13 Comments
matthew runcie
March 19, 2026 AT 21:58This is actually one of the clearest breakdowns I've seen on immunosuppression monitoring. No fluff, just facts. I wish my nephrologist explained it like this.
Thanks for writing this.
shannon kozee
March 19, 2026 AT 23:14CBC, creatinine, magnesium - these are the big three I watch like a hawk. One dip in magnesium and I’m cramping up like I did a hundred squats. It’s wild how such a tiny mineral can wreck your day.
Shaun Wakashige
March 20, 2026 AT 00:59lol i just skip my labs now. 🤷♂️
Thomas Jensen
March 20, 2026 AT 17:53They say TTV is the future but I’ve been told the real reason they’re pushing it is because Big Pharma wants to sell more tests. I’ve got a buddy whose lab results got manipulated to keep him on expensive meds. You think they’d let you off the hook? Nah. They need your money. The system’s rigged. I’ve seen it. And TTV? Probably just another way to keep you hooked.
Johny Prayogi
March 21, 2026 AT 16:57TTV is a game changer. 🚀 My transplant center started using it last year and my infection rate dropped HARD. No more pneumonia scares. I feel like I’ve got a real dashboard now - not just guesswork. If your center doesn’t offer it, demand it.
Natali Shevchenko
March 22, 2026 AT 05:00I think about how fragile we are, you know? One pill, one blood draw, one number on a screen - and your whole body’s balance hangs on it. It’s not just medicine. It’s a quiet dance between your immune system and the drugs that tame it. We don’t talk about that enough. The poetry of survival. The silence between the lab results. That’s where the real story lives.
Solomon Kindie
March 23, 2026 AT 11:19so like if ttv is high then ur immune system is weak right? but then why dont they just stop the meds? like its all just numbers man. we are just meat bags with dna and meds. why do we pretend its science when its just guesswork with fancy machines? lol
Nicole James
March 24, 2026 AT 15:56Wait… so TTV is being pushed… but what if it’s being used to justify MORE drugs? What if the virus is engineered? I read a paper once - not published, mind you - that suggested TTV was a byproduct of mRNA tech. Coincidence? I don’t think so. They’re tracking us. The labs. The numbers. The ‘peace of mind’? That’s the opiate of the masses.
Nishan Basnet
March 24, 2026 AT 18:31I’m from India and we don’t have access to half of this. My cousin got a kidney transplant and they still use immunoassays because LC-MS/MS costs more than his monthly rent. He’s on cyclosporine and his creatinine keeps creeping up. No TTV. No AI. Just prayers and hope. This post is beautiful… but for most of the world, it’s science fiction.
Allison Priole
March 26, 2026 AT 11:48I love how they say monitoring is 'peace of mind' but honestly? It’s the opposite. Every time I see a lab slip, my heart drops. I’m not just checking numbers - I’m checking if I’m still alive. It’s exhausting. I wish we could just… not do this. But I know if I stop, I die. So I keep showing up. Even when I hate it.
Timothy Olcott
March 27, 2026 AT 20:16USA has the best transplants. Europe? Overregulated. India? Barely functional. This is why we lead. TTV, AI, point-of-care - we’re building the future. Other countries? They’re still using slide rules. #AmericaFirst #MedTechSuperior
Desiree LaPointe
March 28, 2026 AT 03:15Oh, how quaint. You think TTV is revolutionary? My cousin, a PhD in virology at Hopkins, told me this was published in 2018. And the AI model? It’s just logistic regression with a fancy name. You’re all celebrating the rebranding of 2015 data. How endearing. The real innovation? The fact that you still believe in 'peace of mind' from a blood test. Darling, you’re not being monitored - you’re being marketed to.
Paul Cuccurullo
March 29, 2026 AT 22:46I have been on immunosuppressants for over a decade. I’ve watched my kidneys slowly decline. I’ve cried in the lab waiting room. I’ve missed birthdays because I was too weak to leave the house. But I’ve also lived. I’ve held my daughter’s hand. I’ve watched her grow. And this - this meticulous, exhausting, expensive monitoring - is the reason I’m still here. Not because of science. Not because of innovation. But because someone, somewhere, refused to let me go. Thank you.